Production and prurification of albumin using bacterial overexpression.

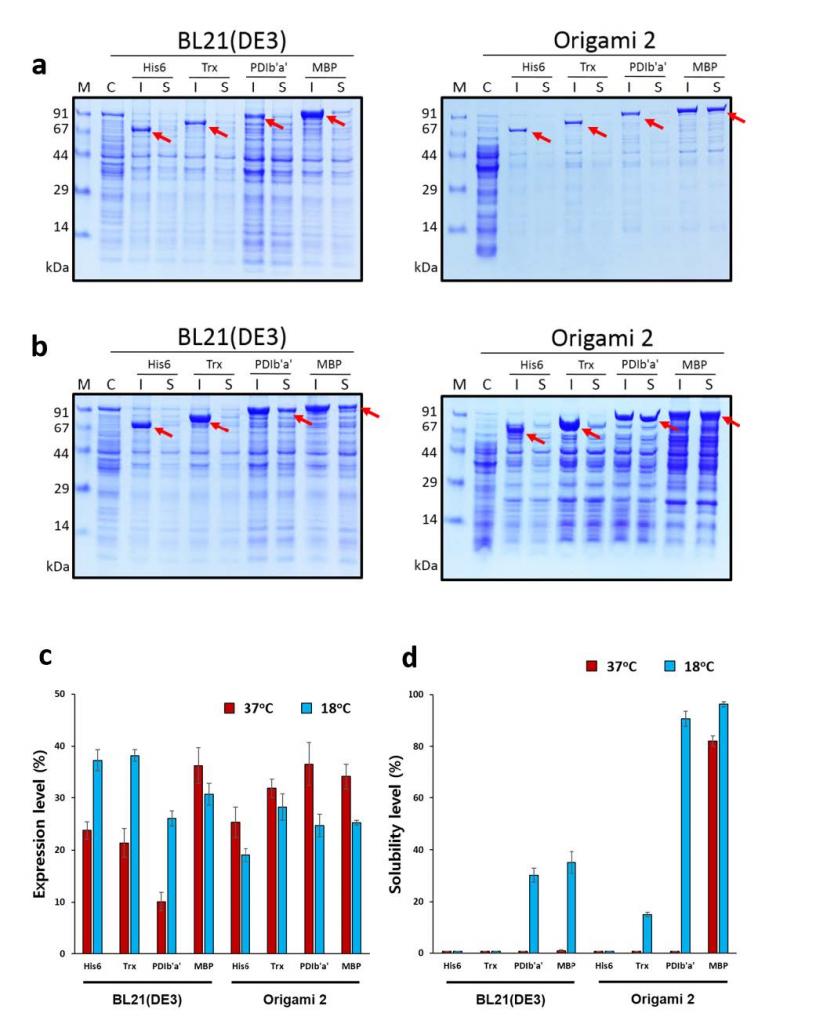
Human serum albumin (HSA), the most abundant serum protein in healthy humans, plays important roles in many physiological processes and has wide clinical and research applications. Despite several efforts to obtain recombinant HSA (rHSA) from bacterial and eukaryotic expression systems, a low-cost and high-yield method for rHSA production is not available. The large molecular weight and high disulphide content hamper the expression and production of rHSA using bacterial hosts. Hence, a strategy that uses a fusion technique and engineered Escherichia coli strains was employed to improve the expression of soluble rHSA in the bacterial cytoplasm. The solubilities of the b'a' domain of human protein disulphide isomerase (PDIb'a')- and maltose-binding protein (MBP)-tagged rHSA expressed in Origami 2 at 18 °C were notably increased by up to 90.1% and 96%, respectively. A simple and efficient protocol for rHSA purification was established and approximately 9.46 mg rHSA was successfully obtained from a 500-mL culture at 97% purity. However, rHSA was mostly obtained in soluble oligomeric form. By introducing a simple refolding and size-exclusion chromatography step, monomeric rHSA was obtained at 34% yield. Native polyacrylamide gel electrophoresis confirmed the similarity in the molecular weights between E. coli-derived monomeric rHSA and commercial monomeric HSA.
The publication has the contribution of Dr. Đỗ Bích Hằng, Lecturer of Faculty of Pharmacy and published on Journal of Protein Expression and Purification volume 167, March 2020. Fulltext could be downloaded from here.







