Systems-level multi-omics characterization provides novel molecular insights into indomethacin toxicity
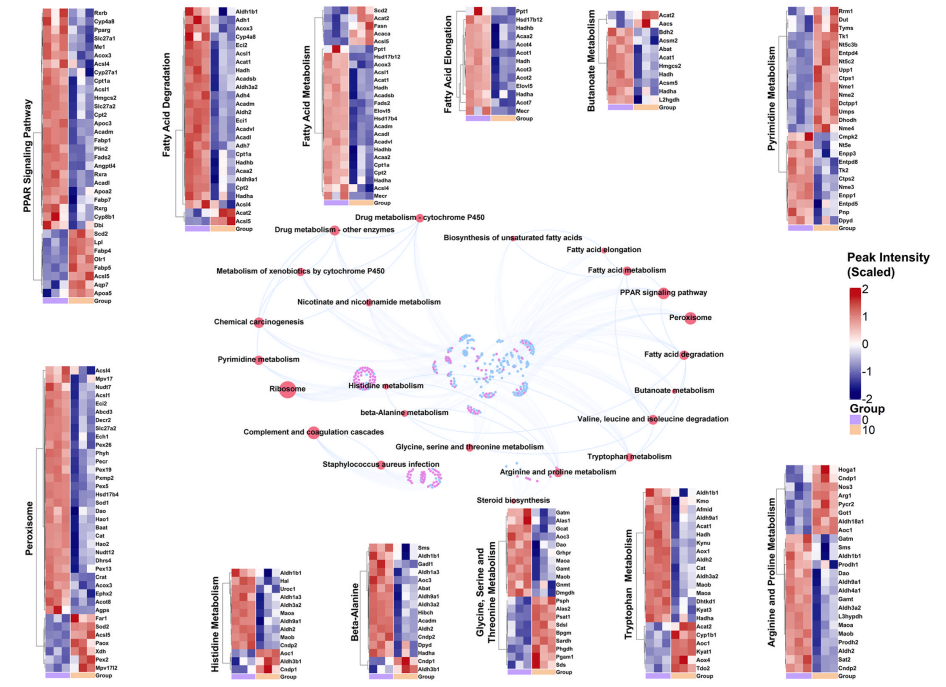
The mechanism of indomethacin toxicity at the systemic level is largely unknown. In this study, multi-specimen molecular characterization was conducted in rats treated with three doses of indomethacin (2.5, 5, and 10 mg/kg) for 1 week. Kidney, liver, urine, and serum samples were collected and analyzed using untargeted metabolomics. The kidney and liver transcriptomics data (10 mg indomethacin/kg and control) were subjected to a comprehensive omics-based analysis. Indomethacin exposure at 2.5 and 5 mg/kg doses did not cause significant metabolome changes, whereas considerable alterations in the metabolic profile compared to the control were induced by a dose of 10 mg/kg. Decreased levels of metabolites and an increased creatine level in the urine metabolome indicated injury to the kidney. The integrated omics analysis in both liver and kidney revealed an oxidant-antioxidant imbalance due to an excess of reactive oxygen species, likely originating from dysfunctional mitochondria. Specifically, indomethacin exposure induced changes in metabolites related to the citrate cycle, cell membrane composition, and DNA synthesis in the kidney. The dysregulation of genes related to ferroptosis and suppression of amino acid and fatty acid metabolism were evidence of indomethacin-induced nephrotoxicity. In conclusion, a multi-specimen omics investigation provided important insights into the mechanism of indomethacin toxicity. The identification of targets that ameliorate indomethacin toxicity will enhance the therapeutic utility of this drug.
The study has the contribution of Dr. Nguyen Truong Huy and Dr. Le Quoc Viet from Faculty of Pharmacy, Ton Duc Thang University.







